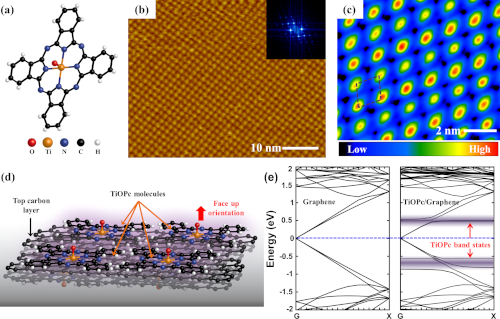
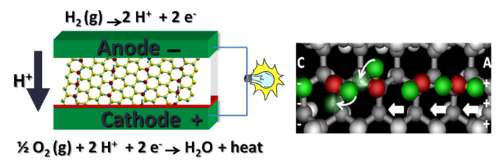
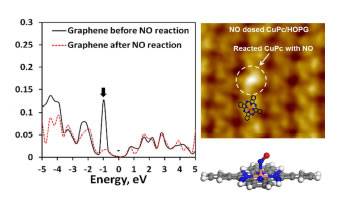
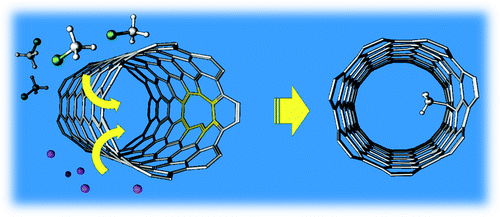
Chemical Engineering Research
Research Areas
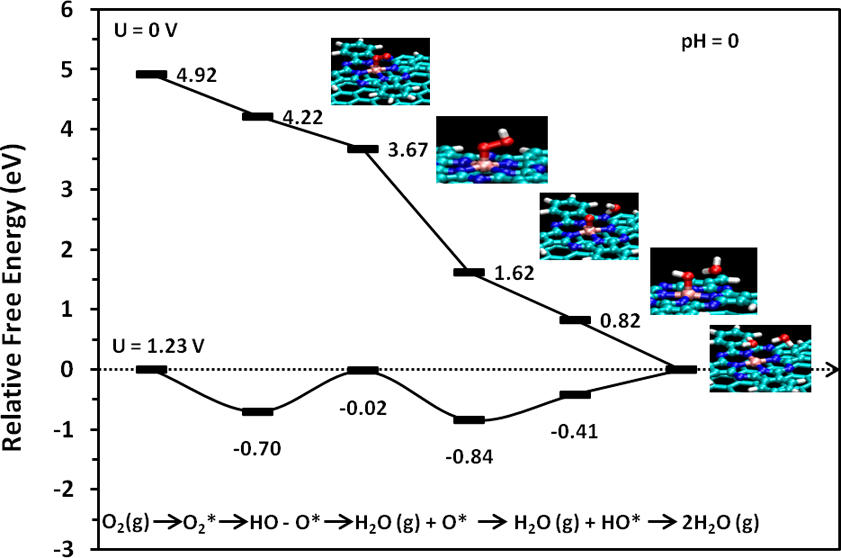
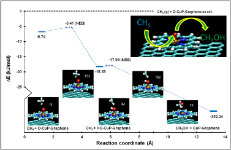
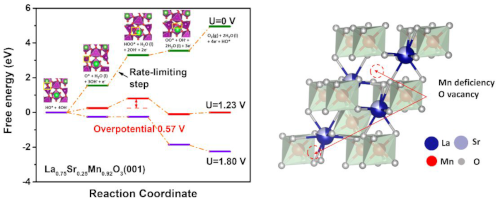
Catalyst Design for Energy and Environment
Additive Manufacturing
Materials for Low-power Electronic and Thermoelectric Devices
Anhydrous Proton Transport Membranes
Graphene As High Chemical Sensing Platform
Sorbent Materials
Funding and Other Resources
![]()

![]()